Types of Documentation Used in GMP Environments
- Published on: Dec 16, 2020
Completed batch records must accurately reflect what actually happened during manufacture and packaging.
Once the product leaves the warehouse, only the batch and test records remain as evidence of good manufacturing practice. If there is an error in the record, particularly if it relates to a formulation, should be formally investigated as soon as possible.
Waiting to see if the GMP auditors find it misses the point, as it may delay recovery or correction of a significant safety problem.
All procedures that may impact product quality, that is, those procedures that would be governed by GMP, must be introduced in a formal manner.
GMP rules on introducing a procedure include:
- Procedures must have an official introduction or “Effective on:” date.
- Procedures must be authorized or approved.
- Procedures must be accompanied by a documented training event for all users. The resulting training record must be available.
- Procedures must be located such that they are directly available to the users.
Specifications:
To ensure consistent quality of materials and product, there are written specifications for all:
- Starting materials
- Packaging materials
- Intermediate products
- Finished products
Specifications need to be approved by the Quantity Department, and are not to be changed without their written approval.
Documents for Starting materials:
The requirements for starting materials must be well-defined and documented to ensure that you get the material you want, and that there are no mix-ups.
Starting materials are defined by:
- A standard name
- The supplier’s code
- A unique item code
- A unique lot/batch number
Specifications for starting materials should include:
- Inspections and/or tests required
- Acceptance criteria
- Storage conditions
- A retest or expiry date
Documents for Packaging materials:
The requirements for control of packaging materials must also be well-defined and documented.
Packaging materials are defined by:
- A standard name
- A unique item code, including a revision code
Specifications for packaging materials should include:
- A detailed description of this item
- Inspections and/or tests required
- Acceptance criteria
- An approved label copy
Documents related to intermediate product:
Specifications for intermediate and bulk products should be available if these are received or dispatched, or if data obtained from tests on intermediate or bulk products are used for the evaluation of the finished product.
The specifications should be similar to specifications for starting materials or for finished products, as appropriate.
Intermediate or bulk products that are purchased should be handled as though they were starting materials.
Documents for finished product:
Specifications for finished products should include or reference:
- An exact statement of the therapeutically active substances
- The product name and code (if any)
- The pharmaceutical dosage form
- Physical appearance
- All tests and limits used to release the finished product
- Details of, or reference to, the test methods
- Sampling instructions
- The shelf life and storage conditions
Documents associated with Inward goods:
Starting materials are purchased only from approved suppliers and to authorized specifications.
Each separate material is given a Unique Identifying Number (UIN) from the Goods Received Register.
The UIN and the standard name are used throughout manufacturing to identify that material and to trace its history from supplier to customer.
- Goods Received Register
- Standard Names List
- Status labels
- SOPs and Work Instructions
Goods Received Register:
A register should be established showing the receipt of starting materials. The register should include:
- Date of receipt
- The standard name of material
- The supplier’s and the manufacturer’s name for the material (if different)
- Manufacturer’s batch or reference
- Total quantity and number of containers received
- The UIN assigned after receipt number
Standard Names List:
A Standard Names List, as a minimum, contains:
- The approved supplier name for the chemical
- The approved manufacturer name
- The company item code
- The company standard name that is used on the Bill of Materials
- The grade of the material, e.g. USP, BP, EP
- Reference to any Material Safety Data Sheets (MSDS) or safe-handling requirements
A list showing the standard name for each starting material should be established.
Standard names should be specific and be designed to minimize mix-ups.
The standard names specified on this list should be used to label and identify starting materials during storage and manufacture.
The standards names should be consistent with the master batch formula.
Status labels:
The status of any material should be evident from the visual appearance of its status label.
Status labels are required for all materials and products, including intermediates.
“Reject” labels should be used only for materials that are unfit for use. Those of uncertain status or destined for recovery, re-processing and the like should be designated “Held” or “Quarantine”.
Standard Operating Procedures (SOPs) and Work Instructions (WIs):
SOPs and WIs are documents that describe how to perform a particular operation or task.
They must be readily available at the worksite.
You must understand them and follow them exactly for all duties you are trained to perform.
Production Documents:
Production documents provide evidence of the processing and quality of products.
All personnel must understand the importance of these documents. Manufacturing instructions must be followed exactly, and batch records must be accurate, be complete, and recorded in real-time.
- Master formula and processing instructions
- Batch records
- Labels and tags
- Logs and forms
Master formulas and processing instructions:
The master manufacturing formula provides a complete list of ingredients and their respective quantities to be used in processing.
Master processing instructions describe exactly:
- The way in which a product is to be manufactured
- The sequence of processing
These instructions must contain enough information to allow the company to re-create the manufacturing operation exactly as it is supposed to occur.
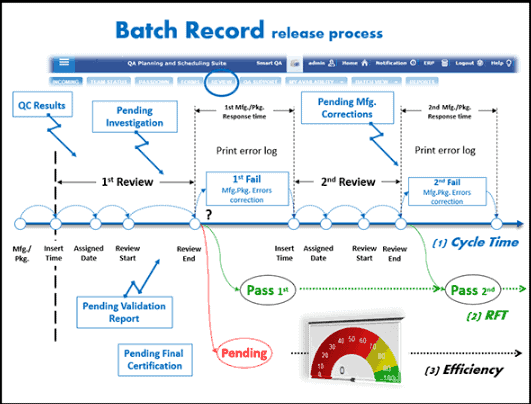
Batch records:
Batch records are properly authorized copies of the master processing instructions.
They provide instructions of what to do and a record of what was done, including a list of all materials and components used and the quantities used.
Batch records provide evidence of the quality of our products because operators are required to sign their name to certify that the operations took place as stated.
Labels and tags:
Typical labels and tags used within the company include cleaning tags and calibration tags.
These labels require (as a minimum) the date of the operation and the signature or initials of the person applying the label.
Logs and forms:
Logs and forms, such as calibration or cleaning records, document and provide evidence that particular operations have been performed.
The information you are required to enter may be a signature stating that the particular function has been successfully completed, or it may be the recording of a particular piece of information that is relevant to the manufacture of a product.
Packaging Documents:
Packaging documentation provides evidence that the right products have been packaged in the right container with right label, that is, the product is correctly identified.
Personnel must ensure that the packaging instructions are followed exactly.
- Packaging Bill of Materials and Master Packaging Instructions
- Packaging batch records
- Logs and forms
Packaging Bill of Materials and Master Packaging Instructions
The Packaging Bill of Materials and Master Packaging Instructions contain all the specific information required for labeling a product. These instructions should also include areas for calculating final yield, and for calculating the reconciliation of packaging items.
Packaging batch records
Packaging batch records are properly authorized copies of the master packaging instructions.
Packaging batch records provide instructions of what to do and are accurate records of what was actually done. They include a list of all materials and components used and the quantities used.
Packaging batch records provide evidence of the product quality because operators are required to sign their name to certify that the operations took place as stated.
Packaging batch records provide evidence of correct product identity. Operators are required to sign their name to certify that the operations took place in line with approved instructions, and the current revisions of the printed matter were used in the packaging operation.
It is standard practice to apply one certified sample of each item of printed matter used in the packaging as part of the packaging batch record.
Logs and forms
Logs and forms, such as calibration or cleaning records, document and provide evidence that particular operations have been performed.
The information you are required to enter may be a signature stating that the particular function has been successfully completed, or it may be the recording of a particular piece of information that is relevant to the manufacture of a product.
Quality Control (QC) documents
QC records provide evidence of the quality of materials and products.
Testing must be performed in accordance with methods described, or referenced, in specifications.
Results of all testing are recorded and retained as part of the batch records.
- Test methods
- Test records
Test methods
Samples of starting materials, in-process goods and finished products are taken in accordance with written sampling plans and procedures.
Testing is performed in accordance with the methods described or referenced in specifications.
Test methods contain, or reference, all the information needed to test materials and products.
Test records
Test records are documents that provide accurate evidence of the quality of products. As such, they are very important legal documents.
The record for a test must contain all testing data, dates and signatures. It must also identify the test method and must allow all calculations to be checked.
The method of recording should allow for reviews of past results and the detection of trends.
Key points about documentation and record keeping:
- A good documentation system is the cornerstone of a GMP compliance system.
- All personnel must follow the written and approved documents in the day-to-day operation of the manufacturing facility.
- It is the responsibility of management to provide a documentation system that conforms to the GMP requirements, and then train the users in the requirements of the system.
- It is your responsibility to understand the documents and use them correctly.
- When you sign or initial a record saying that you carried out a process, you are certifying that the process was carried out according to the written and approved procedures.
- When you sign or initial a checking step in a record, you are saying that you observed what was done, and can verify that it was done correctly.
- Remember: if it’s not documented, it wasn’t done.
It is only by having a documentation system that everyone complies with, that we can demonstrate that we have manufactured a quality product.

Author: Kazi Hasan
Kazi is a seasoned pharmaceutical industry professional with over 20 years of experience specializing in production operations, quality management, and process validation.
Kazi has worked with several global pharmaceutical companies to streamline production processes, ensure product quality, and validate operations complying with international regulatory standards and best practices.
Kazi holds several pharmaceutical industry certifications including post-graduate degrees in Engineering Management and Business Administration.
Related Posts
Pharmaceutical sampling procedures for non-sterile products
Equipment cleaning procedure in pharmaceutical, Do’s and Don’ts
Dispensing Technique in GMP