
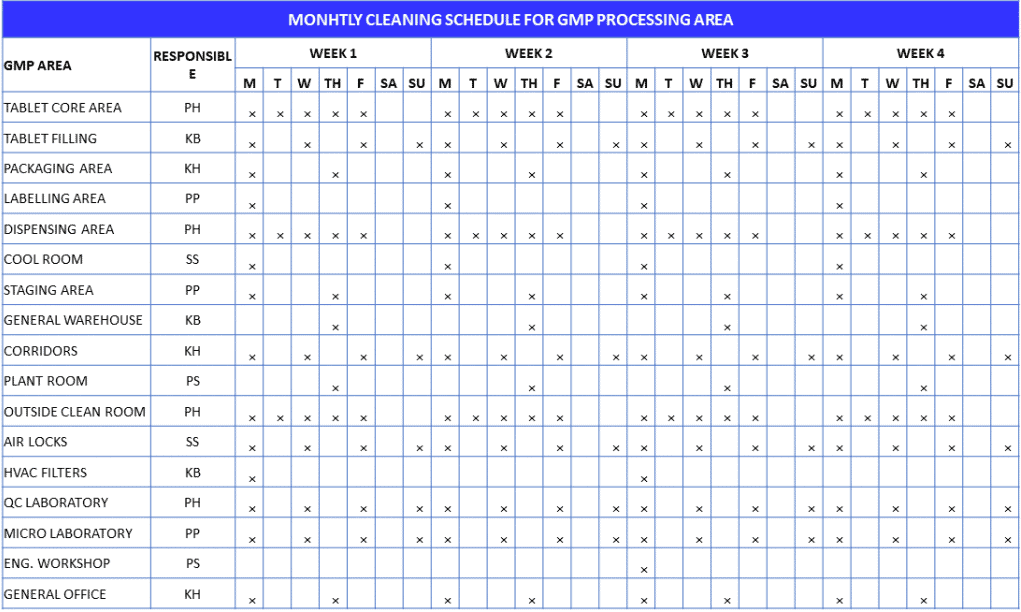
Typical cleaning records produced by pharmaceuticals manufacturing plants
- Published on: Dec 22, 2020
One of the most stated rules in GMP is “You cannot test quality into products. You must build it in at each step in the entire process.” This statement is especially true with regard to effective cleaning and prevention of cross-contamination in products. One of the important process steps is to maintain GMP cleaning records diligently.
No amount of laboratory testing can let you substitute prevention from cross contamination. If you complete cleaning and cleaning records correctly, they should act as evidence to verify that each equipment that come in contact with product or raw materials was cleaned.
Equipment cleaning should be carried out:
– By using validated written procedures
– By the trained employees
– Prior to use and store correctly as per its status tag
Cleaning records must be evidence via inspection and monitoring that the equipment was clean and free from contaminants when processing is commenced. Activities must be recorded what previous product was processed in the equipment prior to the current batch.
What are the guidelines around GMP cleaning records?
According to US FDA CFR 211 – Subpart J-Records and Reports, Sec. 211.182 Equipment cleaning and use log.
– A written record of major equipment cleaning, maintenance (except routine maintenance such as lubrication and adjustments) and use shall be included in individual equipment logs that show the date, time, product and lot number of each batch processed.
– If equipment is dedicated to manufacture of one product, then individual equipment logs are not required, provided that lots or batches of such product follow in numerical order and are manufactured in numerical sequence.
– In cases where dedicated equipment is employed, the records of cleaning, maintenance and use shall be part of the batch record.
– The persons performing and double-checking the cleaning and maintenance shall date and sign or initial the log indicating that the work was performed. Entries in the log shall be in chronological order.
Why maintaining cleaning records is important in GMP facility?
For decades, it has been a basic GMP requirement that all cleaning steps in a pharmaceutical plant be documented accurately and also validated to prove they work. Once the steps are validated, your employees must only follow the approved procedure.
Some kinds of contamination are often difficult to detect by physical inspection. The claim: “It looks clean, therefore it must be clean” is not necessarily true. Laboratory testing for potential contaminants is generally not practical, economical, nor reliable.
You need to employ both cleaning and sanitation practice to reduce potential contamination. Contamination could negatively impact your end product if it is present in the manufacturing facility or on production equipment.
– Chemical contamination
– Environmental contamination
– Biological contamination
Cleaning vs sanitation practice
Cleaning is the removal of visible and microscopic contamination by dirt, extraneous mailer or product residues by mechanical or physical means. During cleaning you should perform visual inspection first and followed by laboratory testing to verify that the cleaning continues to be effective.
Effective cleaning usually requires you to use cleaning agents such as detergents and solvents, which are used under specified conditions of pH, temperature, time and solvent concentration.
A concern with cleaning is whether or not the cleaning agent itself will have a negative effect on the life of the equipment or on the quality of the product.
Sanitation is the reduction of microbiological contamination. You can achieve effective sanitation by the use of chemicals.
However, it can also be achieved by hearing and even vigorous mechanical action, such as scrubbing.
(Note: Cleaning may result in a partial reduction of the microbial load.)
If sanitising chemicals are used, it may be necessary to remove the sanitiser’s residues by further cleaning. It is therefore very important to use the specified amount of sanitising agent.
There are several factors that determine how effective sanitising will be. These include:
– The number and types of microbes
– The type of material containing the microbes
– The concentration, temperature and pH of the sanitising agent
– The length of time you sanitise for
Note: It is impossible to sanitise a dirty surface. The surfaces must first be thoroughly cleaned.
You can find further information on equipment cleaning, facility cleaning, cleaning and sanitation practices, cleaning validation for formulated products and API plant in our other blog articles.
What are the benefits of cleaning records in manufacturing operation?
Cleaning records are critical in a manufacturing operation. They are needed to:
– Provide evidence that equipment has been cleaned and checked before use
– Demonstrate that cleaning was done in accordance with Standard Operating Procedures (SOP).
– Provide traceability and help in problem solving should a product prove to be contaminated
– Cleaning records are one of the real source of information on a batch after it has been released, so they must be accurate and complete
– They provide legal evidence that your company followed GMP rules for cleaning.
– Cleaning records are used to investigate product complaints/adverse events.
– Complaint investigation is a critical activity to protect patients’ health. By maintaining cleaning records accurately will facilitate you to reach correct conclusions of an investigation.
– Cleaning records are also useful to investigate trends or problems.
– They are always reviewed during government audits.
– Auditors review cleaning records to ensure they are accurate, complete and follow validated cleaning procedure. Auditors place importance on this review when forming opinions on company GMP standards.
This is why equipment must have status tags stating “Clean” or “To be cleaned” etc.
The GMP rule is to never use equipment unless it has a “Clean” status label attached, and has been inspected as clean.
210 SOPs, 197 GMP Manuals, 64 Templates, 30 Training modules, 167 Forms. Additional documents included each month. All written and updated by GMP experts. Checkout sample previews. Access to exclusive content for an affordable fee.
What is ideal practice of completing cleaning records:
Here are some simple rules for you while completing cleaning records:
– Records must be accurate.
– They must be completed in real-time at the time of cleaning.
– Records must be legible.
– Records must be completed in a non-water-soluble pen.
– Records must be stored to prevent loss or destruction.
– Never use whiteout or erasers.
– To correct data, cross out the incorrect data with one line, enter the new information, sign or initial it, and date it.
– Fill in all fields, even if you write “Not Applicable”.
GMP rules require that cleaning records are completed as the event occurs.
The reason for this is that if people rely upon memory at a later time, the record may not be accurate. The accuracy of batch records is critical to in-process controls, release of the batch to the market, and for later investigation if there is a customer complaint.
Leaving notes for other staff members is also a problem because they may get lost and are not part of the formal record system.
Good practice while recording cleaning information:
– Only use permanent ink, preferably blue or black to complete a cleaning log.
– You must not use white correction fluid or writing over an entry to correct an error as that is not acceptable.
– If a correction is necessary, cross out the incorrect information using one line.
– Enter the correct information near the incorrect information accompanied by your initials and the date the correction was made.
– If repeated information is required for several lines in a document, e.g., initials, do not use ditto marks (“) or arrows (¯). Initials should be entered each time they are required.
You should record cleaning data directly onto the production batch record, forms, logbook, or worksheet.
Errors, which are corrected based on information contained in another source document, must be referenced or the source document must be attached.
Personnel who verify calculations must do so independently.
Some basic GMP rules around cleaning
The GMP rules are very explicit regarding the requirement to clean & sanitise equipment and the manufacturing facility.
Since most residues cannot be seen nor reliably tested for, your company’s standard procedures and cleaning records are relied upon as evidence of cleaning.
1. Cleaning procedures
GMP requires the cleaning procedures to be fully documented in written procedures. These procedures are initially validated (shown to be effective) specifically for an item of equipment or an area.
Once validated, the procedures are then published and used.
2. Cleaning validation
All cleaning procedures should be validated (shown to be effective) under specific conditions of use. These conditions of use are specified in the written procedure.
If employees alter the conditions of use, the cleaning methods may be “invalidated” and may not be effective.
For example, it would seem logical that adding more sanitiser than the procedure requires would make cleaning faster or better.
This is not always the case. Adding additional sanitiser may alter the pH of the cleaning agent and make it less effective.
The relevant GMP rules are to only clean under validated conditions and to follow procedures exactly.
3. Cleaning conditions
The written procedures describe the required conditions under which cleaning is optimal. These conditions may include a range of the following:
– Strength of the cleaning or sanitising agent (e.g. 2.0% v/v)
– The type of water or solvent to be used
– The pH of the cleaning agent
– The temperature of the water to be used
– How much to dismantle equipment before cleaning commences?
– The contact or residence time for the sanitising agent on the surface
– What agents to use to clean off the sanitising agent
– What standard of water to use in the final rinse (for example, purified or water for injection as the final rinse)?
4. Cleaning records
One essential GMP rule is the keeping of detailed cleaning records. Cleaning records provide not only proof that cleaning took place, but also provide evidence of the cleaning outcomes.
For example, that the surfaces are visually clean or the results of rinse water tests. The records must also identify who did the cleaning, when and must be signed.
For automatic cleaning procedures such as clean in place (CIP), the cycle conditions are usually automatically monitored and the conditions recorded.
Cycle conditions may include the temperature, flow rate, time, concentration of solvent and solvent agitation time. Often CIP systems have alarms when something goes wrong.
The completed and signed records should be attached to the records, since they provide the only real evidence that cleaning has occurred.
What must be cleaned?
The facility must be regularly cleaned. Particular emphasis is placed on areas or rooms where product is processed.
Particular emphasis is placed on the effective cleaning of all equipment in contact with product, and must be cleaned according to validated and detailed procedures.
All equipment in processing areas not in contact with product needs regular cleaning in order to prevent build-up of dust and dirt.
Importance of status tags and cleaning logs in cleaning records
Status tags are essential for communicating to all operators regarding the cleaning status of equipment and rooms. This is especially important when different shifts are sharing equipment.
The status of equipment should be apparent at all times, including during storage.
For example, this status tag on a vessel indicates:
– Whether the equipment is clean or requires cleaning
– When the equipment was cleaned or is to be cleaned
– The previous product in the vessel
– When the equipment is due to be re-cleaned
After it is cleaned, the equipment should be stored with a protective cover to prevent contamination on storage.
Regardless of its cleaning status, equipment should always be examined before use to verify that it is clean and suitable for use.
The images are examples of a status tag as equipment cleaning record. Each requires the date of cleaning, the name of the previous product and the name of the person who did the cleaning. The status tag clearly shows the cleaning status.
You need to create cleaning logs which are essential to ensure that each cleaning task is performed correctly. The logs may detail:
– The area or piece of equipment being cleaned
– The cleaning equipment that is to be used
– The frequency of cleaning
– The responsibilities for the cleaning
Cleaning logs also allow for personnel signing that the cleaning has been performed, and perhaps in some cases, verifying that the cleaning was adequate.
The most important requirement of “FDA 21 CFR 211.182 Equipment cleaning and use log” is that each item of common use equipment must have an individual log that shows batch traceable cleaning history that is independent of the batch record.
How clean is clean?
A commonly-asked question is “How clean is dean?”
The answer is not straightforward. Company SOPs will specify the definition of dean, and appropriate acceptance criteria or limits. Generally, “how clean is clean” depends on a number of factors, including:
– The method of cleaning
– The nature and use of the equipment being cleaned
– The type of products being manufactured (e.g. injectable products, creams)
Some criteria for cleanliness can be:
– Visually clean
Visual inspection should always be used irrespective of other methods employed. Visual inspection should occur both post-cleaning and again before next use of the equipment.
– <0.1% detected
This limit is sometimes used as a default limit because it is less than 1/1000 of the previous active chemical in the next batch.
– Certain per million detected
This limit is similar to the <0.1% limit. A general guide for this limit is not more than 10ppm carryover to the next product.
– No product detected
Sometimes this limit is used when the allowable carryover of residue is higher than the limits of the analytical method being applied.
– No microbial activity detected
This limit is applied to microbial residues post-sanitation. While there is no specific limit required in regulations, the limit chosen should reflect that product risk. For example, for sterile products the limit is nil, while for tablets it may be higher.
Summary
Cleaning and sanitation programs are an important part of GMP, as they protect products from environmental contamination and cross-contamination with other materials or product. These programs are also designed to protect us from the effects of hazardous materials.
One essential GMP rule is to keep detailed cleaning records. Cleaning records provide not only proof that cleaning took place but also provide evidence of the cleaning outcomes.
Cleaning records are critical in a manufacturing operation. In addition to create evidence that equipment has been cleaned and checked prior to use, cleaning records are beneficial prove the cleaning procedures are effective through laboratory testing, assist in problem solving should a product prove to be contaminated, they provide real evidence while investigating product complaints/adverse events.

Author: Kazi Hasan
Kazi is a seasoned pharmaceutical industry professional with over 20 years of experience specializing in production operations, quality management, and process validation.
Kazi has worked with several global pharmaceutical companies to streamline production processes, ensure product quality, and validate operations complying with international regulatory standards and best practices.
Kazi holds several pharmaceutical industry certifications including post-graduate degrees in Engineering Management and Business Administration.
Related Posts
Chemical or Biological Spill Response Procedure
Corrective and Preventive Action (CAPA) Procedure for GMP
Quality Practices in Pharmaceutical Quality Control Laboratory










Is it necessary to record ph and conductivity of final rinsate of an equipment/ parts like clamps , gasket etc for every time we clean ?